|
This is also a possible resonance structure, although the octet rule is not satisfied. The formal charges for all atoms are zero for structure 4, given earlier. 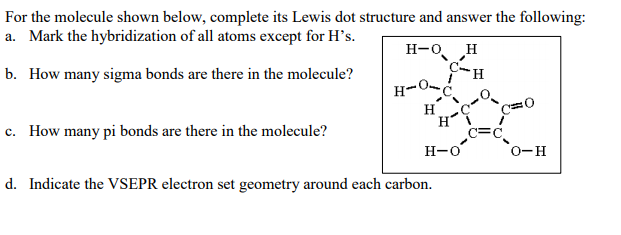
The F axial-I-F axial is actually 168°.\) make structures 2 and 3 more stable or more important contributors. Due to repulsion between the axial F atoms and both the lone pair and double bond, we should expect the F-S-F bond angles to be compressed. With the more repulsive lone pair and the strongest equatorial repulsive force being between the double bond and lone pair, we should expect the F equatorial-I-O bond angle to be less than the 120° angle expected for the parent geometry ( it is actually much less, at 98°). Count the total number of electron domains around the central atom. We use the electron domain geometry to help us predict the molecular geometry. To determine the shape of a molecule we distinguish between lone pairs and bonding pairs. This results in a seesaw molecular geometry. The molecular geometry is the arrangement of the atoms in space. The lone pair and double bond are most repulsive, and should occupy the less crowded equatorial positions rather than the more crowded axial positions. There is one lone pair, a double bond to O, and three single bonds to F atoms around the central I atom. Seesaw geometry is a form of molecular geometry in which the central atom has one lone pair of electrons and is coupled to four bonding groups. This molecule has five electron groups (steric number 5) with an approximately trigonal bipyramidal electronic (parent) geometry. However, molecular structure is actually three-dimensional, and it is important. The molecular geometry is a distorted tetrahedron. Thus far, we have used two-dimensional Lewis structures to represent molecules. As a general rule, lone pairs are slightly more repulsive than multiple bonds, and so we might expect the O-Xe-F bond angles to be 109.5° (and the actual bond angle is 120°). The lone pair is in an equatorial position offering 120 and 90 degree bond angles, compared to only 90 degree bond angles if placed at the axial position. VSEPR theory predicts F-Xe-F bond angles of 90°. The seesaw shape maximizes the bond angles of the single lone pair and the other atoms in the molecule. The result is a square pyramidal molecular geometry. The double bond and lone pair will be directly opposite to each other, designated as axial positions. Question: What is the molecular geometry of SF5- (SHOW WORK, DRAW THE LEWIS DOT STRUCTURE) seesaw linear trigonal planar bent tetrahedral trigonal pyramidal. Valence Shell Electron Pair Repulsion (VSEPR) is a theory that states that the 3d orientation, also known as the molecular geometry, of a molecule is not dependent on its chemical formula but on the repulsion of valence electrons. There is a double bond to O and a lone pair, both of which are more strongly repulsive than the single bonds to F. 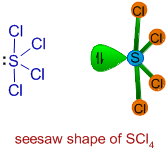
This molecule has six electron groups around the central Xe atom (steric number 6), and thus has an approximately octahdral electronic (parent) geometry. 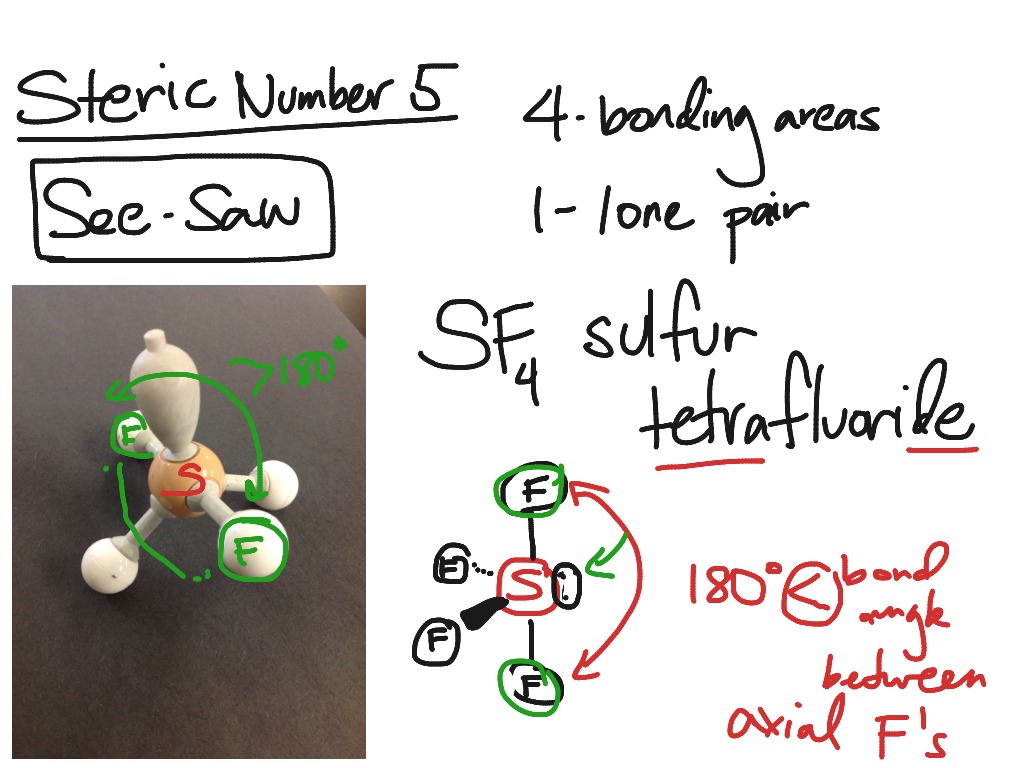
Question: What is the molecular geometry of SF5- (SHOW WORK, DRAW THE LEWIS DOT STRUCTURE) seesaw linear trigonal planar bent tetrahedral trigonal pyramidal trigonal bipyramidal octahedral square planar. Use VSEPR theory to predict the geometries and draw the structures of the following. This problem has been solved Youll get a detailed solution from a subject matter expert that helps you learn core concepts.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
